A new greener chemical method for transforming native sugars
A team of scientists from the Rosalind Franklin Institute and National University of Singapore have developed a more efficient and sustainable chemistry for connecting sugars to diverse partner molecules in a way that mimics nature. Complex sugars perform a variety of jobs within our bodies dependent on the exact molecules that they are attached to and so our ability to attach these sugars has implications for both understanding the function of biology and for producing new drugs.
This research was published today in the journal Nature.
Glycosylation is the process by which sugars are added to partners to make a range of sugar compounds known as glycosides. In our bodies, glycosylation plays widespread roles, including metabolism, trafficking and immunity– when this goes wrong it can be both a hallmark and cause of disease. Therefore, a better understanding of glycosylation will allow us to produce more sugar-based pharmaceuticals that could tackle inflammatory diseases, cancers, and diabetes.
Senior author Professor Ben Davis, Science Director for Chemistry at the Rosalind Franklin Institute and Professor of Chemical Biology at the University of Oxford, said: “Our group has spent a number of years exploring the idea of inserting information into biological molecules such as proteins and sugars to alter their function. One of the things that we have found is that carbon-centred radicals are wonderfully useful yet benign reactive intermediates for achieving new chemistries in biological systems. In this research, by generating free glycosyl radicals as intermediates directly from native sugars we are trying to mimic what is happening in biology to make the synthesis of these glycosides and glycoproteins far more efficient. This efficient ‘harvesting’ of native sugars that can be plugged directly into new glycoconjugates has the potential to open up a number of different avenues including the development of diverse sugar-based therapeutics.”
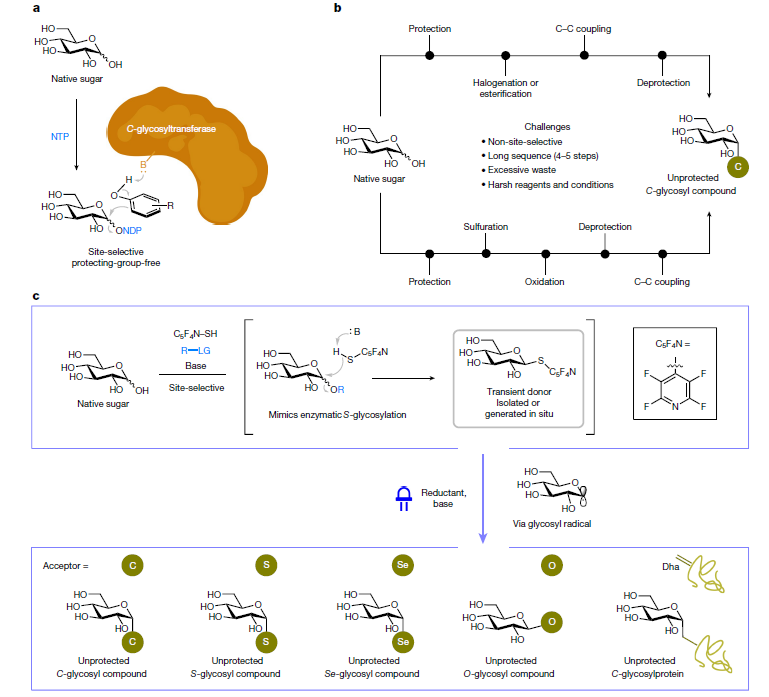
Before now, the methods for producing these glycosides in the lab involved multiple steps, with each step producing waste solvents and often involving harsh conditions or reagents. These steps were required because there are multiple reactive points on the sugars and these would have to be protected or masked to stop unwanted reactions occurring. These inefficiencies have, so far, limited the utility of these glycosides.

Dr Yi Jiang, Postdoc at the Rosalind Franklin Institute and former PhD student at the National University of Singapore, said, “I was really driven to find a new way of doing this process during my PhD, because using the protecting-group strategy was so tedious and I thought there must be a better way of doing this.
“I think the most important thing about this research is that we have been able to showcase the robustness of this approach to directly modify these native sugars. It is a very straightforward method to get useful compounds.”
The research team have now developed a new approach which is free of protecting groups and mimics the natural enzymatic method of creating unprotected reactive intermediates in one step. This method uses a “cap and glycosylate” approach that exploits the harvesting of native sugars – they first activate and substitute the anomeric hydroxyl group on a native sugar to produce an intermediate (cap). Then, when exposed to blue light, the intermediate in the presence of an electron donor and base will react to form the desired glycosides and glycoconjugates (glycosylate). In this paper, the researchers were able to synthesise C-glycosyl, S-glycosyl, Se-glycosyl and O-glycosyl compounds and even synthetic glycoproteins in water to demonstrate the strength of this approach.
Dr Jiang added that “The expertise of our colleagues at the Franklin, Dr Nikita Levin and Professor Shabaz Mohammad, was critical at this stage to elucidate the structure and confirm that this site-specific modification had been made.”
Associate Professor Ming Joo Koh from the Department of Chemistry, National University of Singapore, summed up the benefits of this research, “C-glycosyl compounds especially are interesting carbohydrate mimics which have already been shown to produce potent sugar-based drugs and skincare products. This more efficient method, which produces less waste, should increase research in this field.”
