Franklin researchers demonstrate potential of llama antibodies as a treatment for SARS-CoV-2
During the Covid-19 pandemic, the Franklin team were able to generate nanobodies which are capable of neutralising SARS-CoV-2, and with partners, at UK Health Security Agency (formerly Public Health England) and the Universities of Oxford and Liverpool, were able to prove their effectiveness in an animal model when administered as a nasal spray. The team are continuing to work to produce a robust pipeline capable of creating neutralising nanobodies for novel respiratory viruses.
Background
Nanobodies are single domain antibodies derived from the unique heavy chain only immunoglobins of camels, llamas and alpacas. Their small molecular size, high affinity and stability combine to make them unique targeting reagents with multiple applications in the biomedical sciences, including super-resolution microscopy and non-invasive imaging.
When the Covid-19 pandemic first started, the team at the Franklin quickly saw the potential of nanobodies as a potential theragnostic and pivoted their research to try and address this new crisis.
Antibody therapies have been in use for several decades with monoclonal antibodies, laboratory-engineered proteins, developed to combat viral infections since the emergence of this technology. While monoclonal antibodies share similarities with nanobodies, the nanobodies possess a simpler structure that allows for cloning in Escherichia coli (E. coli), significantly streamlining production. In contrast, monoclonal antibodies require cloning in mammalian cells, resulting in a more complex and costly manufacturing process.
Nanobodies also can be delivered directly to the airways through a nebuliser or nasal spray, so can be self-administered at home rather than needing an injection, giving them a further advantage.
This lab-based product which can be developed quickly, made on demand having considerable advantages and could be used earlier in the disease where it is likely to be more effective.
Science in detail
Researchers at the Franklin have isolated of nanobodies that bind to the spike protein of SARS-CoV-2 by screening nanobody phage display libraries previously generated from llamas immunized with spike proteins from several variants of the virus. The structure and binding properties of these nanobodies have been characterised in detail providing insight into their sites of interaction on the spike protein.
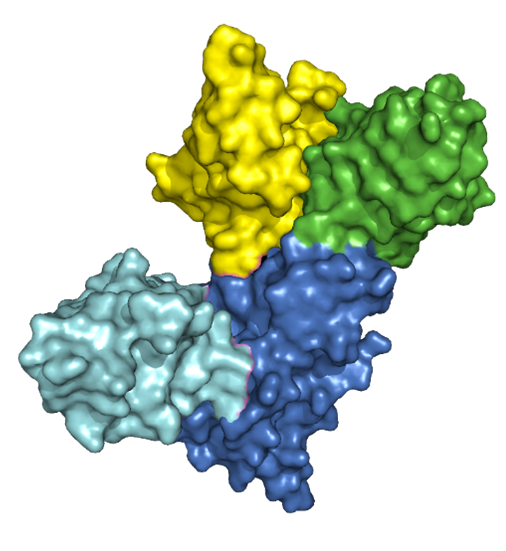
These nanobodies, assembled as trimers to increase binding avidity, neutralized SARS-CoV-2 variants of concern including Omicron strains, in vitro. Working with partners at UK Health Security Agency (formerly Public Health England) and the Universities of Oxford and Liverpool, the team tested the effectiveness of the nanobodies in the hamster model of SARS-CoV-2. They found that they were very effective – UK Health Security Agency described the research as having ‘significant potential for both the prevention and treatment of SARS-CoV-2’, adding that llama-derived nanobodies ‘are among the most effective SARS CoV-2-neutralising agents we have ever tested’. Thus, either alone or in combination could serve as starting points for the development of new anti-viral immunotherapeutics.
For this work the team won the Royal Society of Chemistry’s Chemistry Biology Interface Division Horizon Prize 2022. The Horizon Prizes are for teams or collaborations who are opening up new directions and possibilities in their field, through ground-breaking scientific developments.

The collaboration with UKHSA, Liverpool and Oxford enabled further detailed, structural understanding of the interactions between the virus and neutralising nanobodies, this led to development of a new generation of SARS-CoV-2 neutralising nanobodies that are much more resistant to “escape” by the evolving variants.
Continued mutation of the SARS-CoV-2 poses a significant challenge to the development of effective antibody-based treatments as immune evasion has compromised most available immune therapeutics. Therefore, in the ‘arms race’ with the virus, there is a continuing need to identify new biologics for the prevention or treatment of SARS-CoV-2 infections. The next step will be to combine the nanobodies either as a mixture or physically joined together since it is less likely that the virus will escape from two or more different nanobodies at the same time.
Impact
The US-based biotech company IntegerBio licensed the nanobody technology and were using it to support their efforts to develop new theragnostics against viral diseases including SARS-CoV-2. They are backed by venture capital firm Samsara BioCapital.
Atul Saran, Chief Executive Officer at IntegerBio, says: “The Franklin has been at the forefront of scientific research, and it has been an excellent experience to be able to collaborate with the team. We hope that our capabilities, paired with those at the Franklin, in time, will benefit patients around the world.”