Understanding these first steps along disease progression is important in designing interventions. The group is currently studying a range of neurodegenerative conditions, including proteins implicated in Alzheimer’s Disease and Parkinson’s Disease.
Structural Neurobiology
Developing cutting edge techniques to image structures in the brain on the molecular scale so we can identify early structural changes leading to disease onset.
Errors in neuronal proteins lead to disease. It is important to study structures of these proteins in as close as possible to their native state and in contexts that can recreate potential disease states, i.e. in genetically modified or patient-isolated systems.
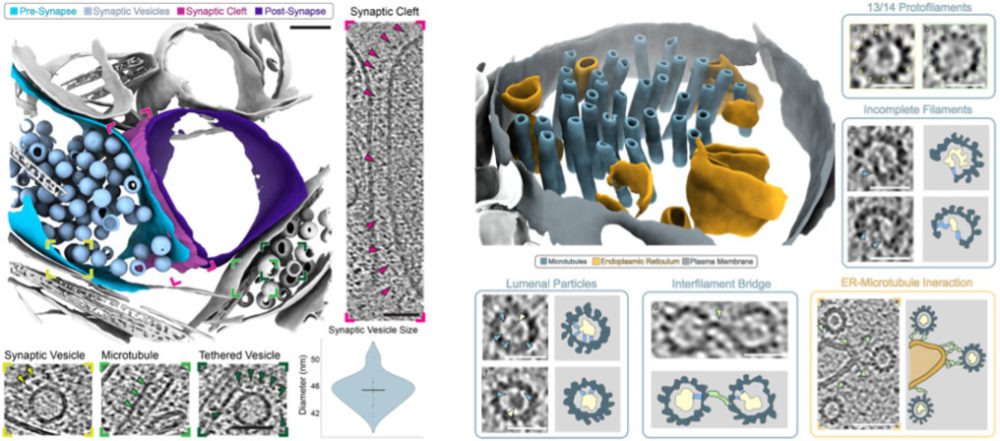
Focused ion beam milling combined with electron cryo-tomography (cryoET) can unravel the molecular details of processes within cells and tissues, ultimately leading to an understanding of how changes in the structures of macromolecules in their native context impact on cellular function.
The formation of neuropathic phenotypes in-situ is poorly understood. We are developing approaches to investigate the morphological and structural changes that occur within brain tissues during the development and onset of neurodegenerative disease using cryoET. The formation of inclusion bodies and neurofibrillary tangles are well-known and prominent features of late-stage disease; however, we aim to understand the molecular changes that occur in proteins that give rise to these features at different stages of disease in-situ. We are developing approaches to image some of these molecular details in human patient derived samples, so that we can best understand what early disease looks like.

Dr Michael Grange
Tomography Group Leader

Dr Calina Glynn
Senior Postdoctoral Research Associate

Dr Charlie Lovatt
Postdoctoral Research Associate

Victoria Garcia-Giner
Postdoctoral Research Associate

Becky Csöndör
PhD Student

Jake Smith
DPhil Student, University of Oxford

Maria Sanita
Research Assistant

Helena Watson
PhD student

Dr Eva Maria Soriano-Jerez
Postdoctoral Research Associate
Thermo Fisher Scientific
Researchers at the Franklin have been working closely with Thermo Fisher Scientific through our Wellcome funded ‘Electrifying Life Sciences’ programme to develop the next generation of electron microscopes for imaging the smallest structures of cells in intact tissues, bring these tools to market and establish new methods and standards for the community to enable their wide use.




