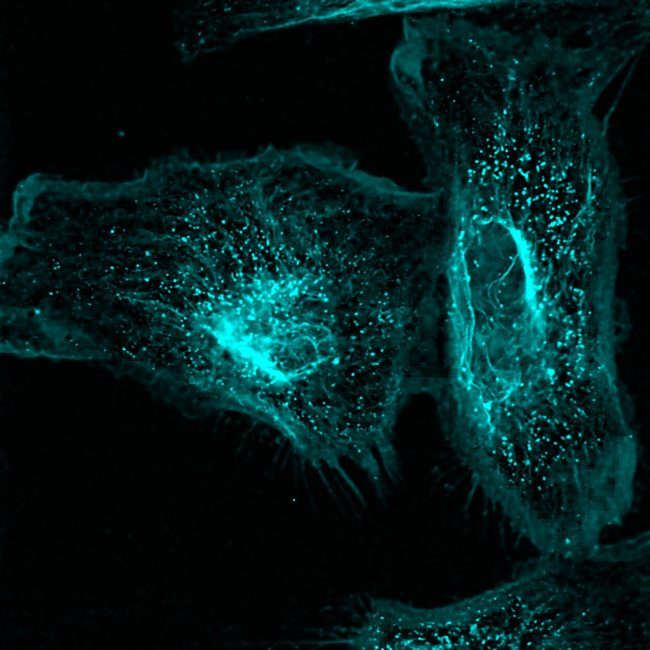
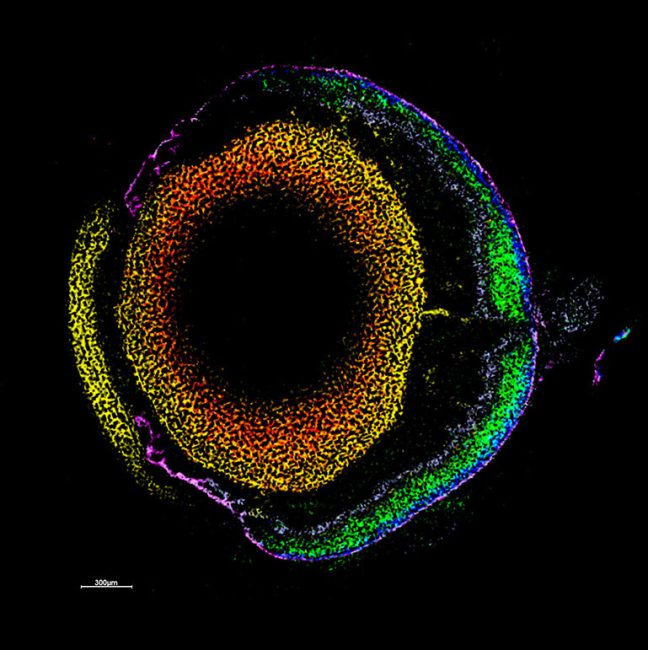
“The Franklin is giving us the time and expertise to dream big and create technologies to look more closely at life at a molecular level. We are developing the novel instruments and techniques which will enable otherwise impossible experiments.”
The ambition
We will work with industry and academia to create novel scientific instruments that will revolutionise our ability to image and understand biological molecules. When these instruments are complete, we expect the wider scientific community to use them for their future research.
Imaging techniques that are being developed now for research may also find applications in the clinic for diagnostic or therapeutic purposes.

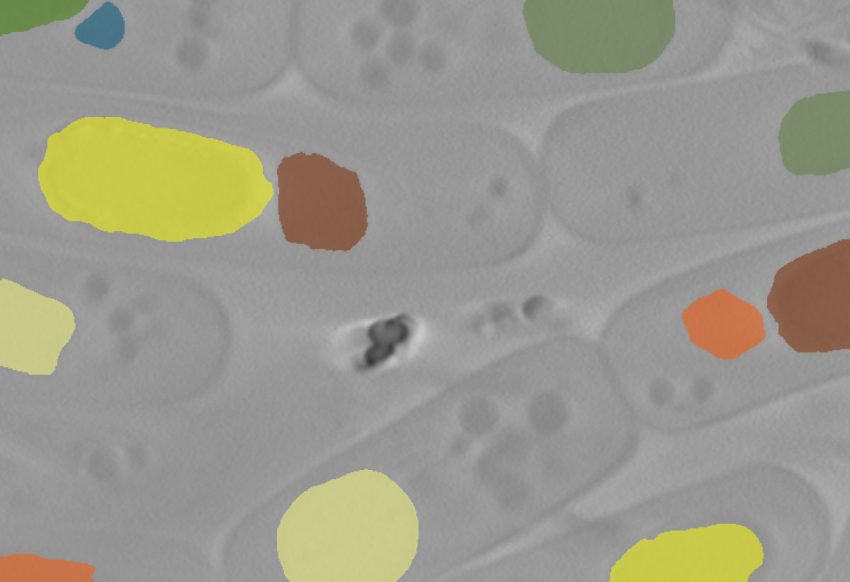
Why?
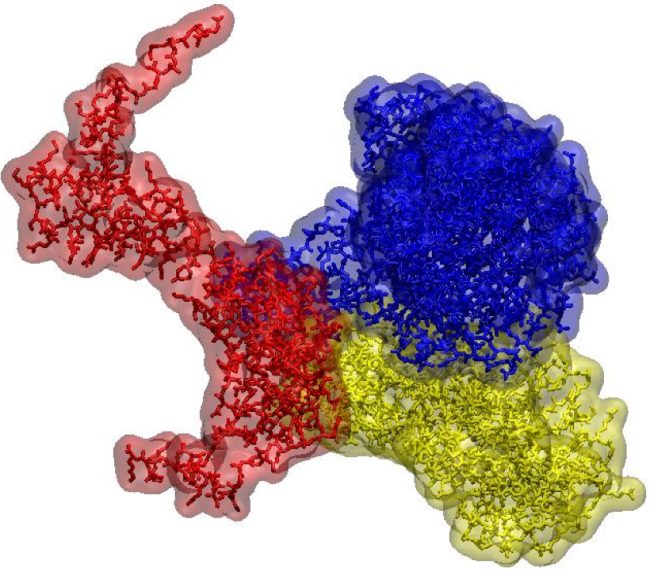
We will be able to determine structures of a broader range of potential therapeutic targets, in biologically meaningful contexts that allow dynamic views of their interactions.
In turn this will provide valuable new insights into how amino acids, proteins, sugars and lipids function in a healthy system and how their malfunction may contribute to disease. The ability to see interactions between biomolecules is vital as many imaging techniques are static whereas, in life, biomolecules move and interact constantly.
What are we doing?
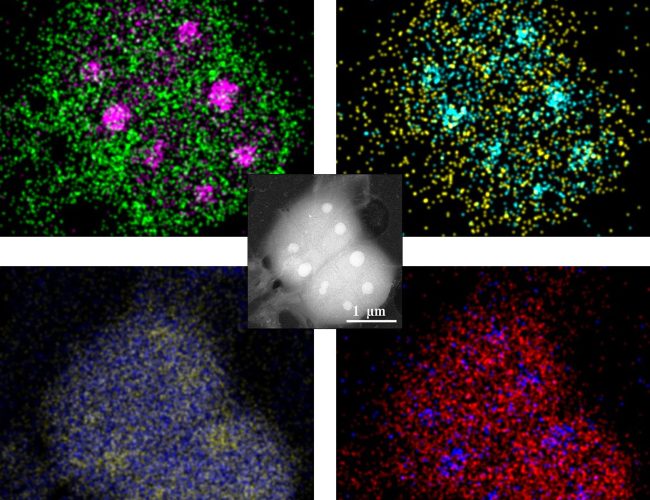
We are developing new imaging technologies and instruments that can detect low levels of multiple signals combined with innovative sample preparation.
We will use new phase sensitive methods for efficient determination of the structures of small (<60 kDa) proteins within fields of view of order 10μm or larger in 2-dimensions and 3-dimensions. These methods include a variety of ptychographic approaches alongside other data acquisition and processing geometries.
In parallel, we will develop automated, autonomous pipelines to enable routine application of these currently complex manual methods.
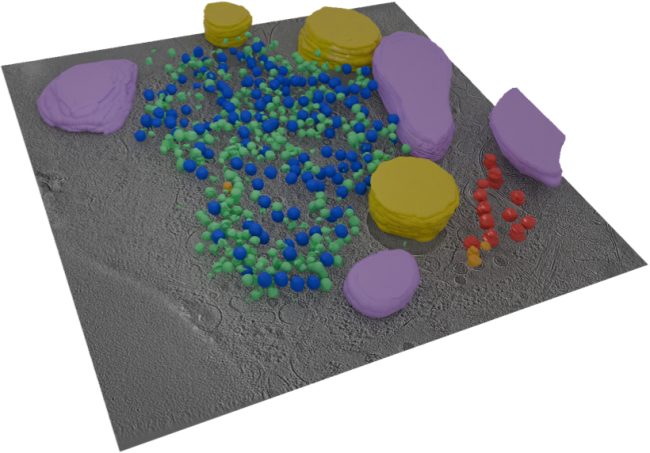
A new range of biological systems will be studied using our patented technologies for graphene-based liquid cells. We will make use of new fast efficient electron detectors to allow dynamic events to be recorded. In combination, these will enable routine large molecule imaging in near native, fully hydrated environments.
In different experimental configurations, the advanced detectors also will make collection of electron diffraction data feasible for structure determination of micro-quantities of hard to crystallise proteins.

Why the Rosalind Franklin Institute?
Our long-term collaboration with JEOL has developed the advanced electron optics that are central to the new instruments including Chromatic aberration correction for the first time in the UK.
Our work in developing new scanning strategies in the SEM for avoidance of charging artefacts was enabled by the close working relationships developed in the Franklin for volume EM imaging . We are actively pursuing a multimodal pipeline involving mass spectrometry and MicroED for high throughput structure determination of small molecules. Our detector developments have involved collaborations with Quantum Detectors (a UK SME located on the Harwell Campus) and STFC.