“These technologies could provide a new era of biological chemistry – helping us to more fully understand the functions of cellular workhorse molecules such as sugars, proteins and lipids.
This is highly ambitious research which could also have unlimited real-world applications in biology and medicine.”
The ambition
This level of molecular manipulation in context in vivo is in its infancy and, so far, has mainly been attempted in vitro. If these techniques could be adapted to work in living systems, they could be a game changer to understand biology and create new diagnostics and therapeutics.
What are we doing?
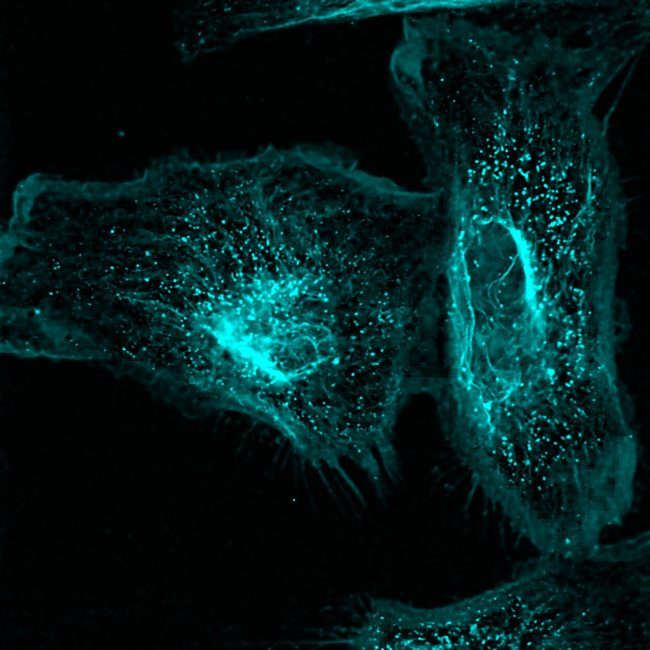
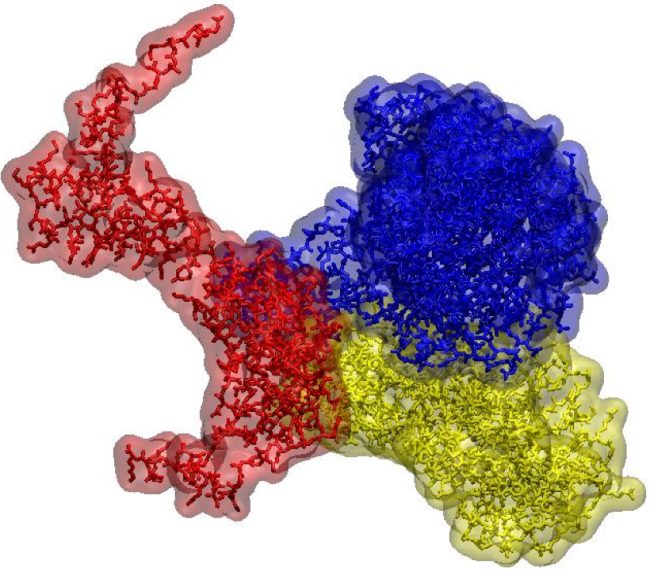
We are developing techniques to selectively manipulate the structure of molecules that ‘do the work’ in living systems, such as proteins, sugars and lipids in vitro and in vivo.

Why?
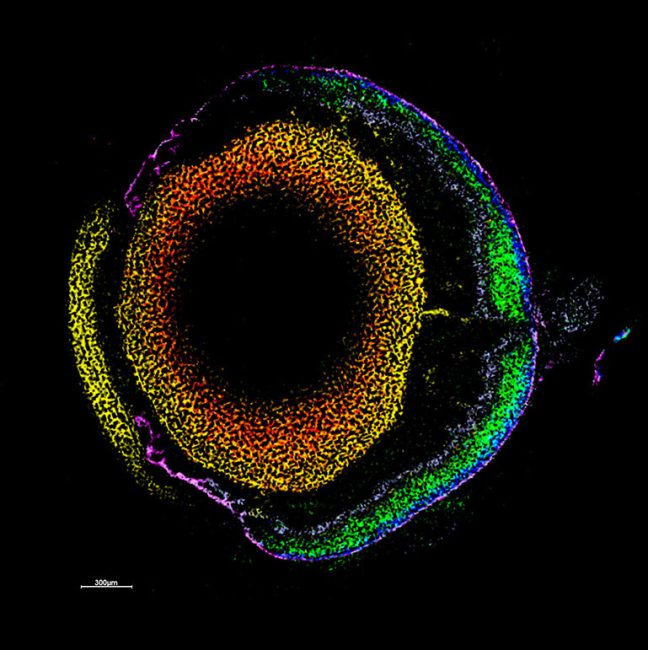
Making specific structural changes to these molecules in the place that they are working gives us unique insights into their cellular functions. These provide greater understanding of biology at a molecular level. We are using these approaches to gain insight into important cellular processes and how pathogens infect cells.
The techniques developed may create new classes of therapies & biotechnologies and enable new ways of visualising and diagnosing pathology. Applying in vivo covalent chemistries as treatments is a bold goal – they represent new modes of treatment, creating fresh possibilities for tackling hard-to-treat diseases.
Research examples
We use organic chemistry to edit biomolecules in very specific ways. A current project is to use biological chemistry to manipulate cell surface carbohydrates to understand their role in cellular communication, pathogen entry and autoimmune conditions.
In the longer term, we hope to be able to use organic chemistry to modify a particular molecule, in a specific organelle, in a specific cell in living organisms. Developing these specific technologies in complex living systems might take decades and will involve novel techniques.

Why the Rosalind Franklin Institute?
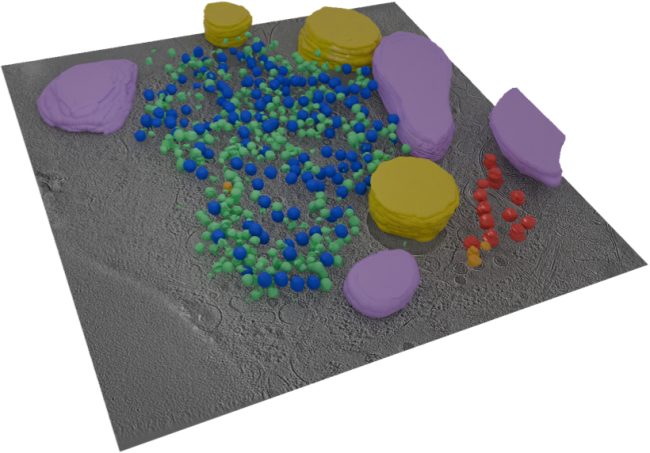
We have access to leading-edge mass spectrometry cryo-electron tomography, and imaging technology equipment and expertise. We are bringing together a team of researchers who have a wide breadth of knowledge about biology and chemistry to solve difficult scientific problems to address this challenge.